At a similar time, endogenous copies of murine leukaemia virus (MLV) were discovered in laboratory mice by two independent laboratories and of avian leukosis virus (ALV) in domestic fowl by Robin Weiss in London.
The association of endogenous retroviruses with cancer was becoming ever clearer and the discovery of oncogenic retroviruses in mice led to much speculation that similar retroviruses may have been causing cancer in humans. This inspired a thorough, yet fruitless quest in the 1970s for equivalent human “tumour viruses”. The interest diminished in the 1980s, with some scientists referring to the elusive targets as “rumour viruses”.
Unbeknownst to the investigators in the mid-1980s, some of the detections of MMTV DNA in human breast cancer tumours may have been copies of the human endogenous retrovirus K (HERV-K) group, which includes the most recently acquired HML-2 (human MMTV-like-2) subgroup, so named due to its sequence similarity with MMTV.
While human endogenous retroviruses have lost the ability to copy themselves and therefore the ability to cause cancer by insertional mutagenesis, the potential to cause cancer through this mechanism still exists for non-LTR elements. Through retrotransposition, ‘jumping’ non-LTR elements can cause genomic instability. Their insertion into the genome can disrupt or alter the function of important genes, regulatory regions, or chromosomes. This genomic instability could contribute to the development of cancer by promoting the accumulation of additional mutations.
A complex relationship with cancer
Even retrotransposable elements that are no longer able to ‘jump’ may still contribute to the development of cancer or affect its progression.
Retrotransposable elements are not normally permitted to transcribe RNA as a result of epigenetic repression. This repressed state is, however, reversible and often lost in cancer cells, where certain retrotransposable elements become transcriptionally activated, with potentially important consequences. These newly activated elements have the potential to, for example, activate oncogenes or tumour-promoting genes within which they have been integrated. They can also influence the epigenetic regulation of adjacent genes. Aberrant retrotransposable element activity can lead to changes in DNA methylation patterns and histone modifications, which can impact the expression of nearby genes and contribute to cancer development.
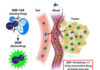
Retrotransposable elements can even trigger immune responses, just like those seen in genuine viral infection. The transcription of retrotransposable element-derived sequences in the genome and subsequent translation into proteins can activate the immune system, leading to chronic inflammation. Prolonged inflammation can create a microenvironment conducive to the development and progression of cancer.
However, once again it is a complex relationship, and conversely, by triggering innate and adaptive immunity to their replication intermediates and protein antigens, reactivated retrotransposable elements can enhance the immunogenicity of cancer cells, leading to more effective anti-tumour immune responses.
It is worth noting that not all retrotransposable elements have the same impact, and the role of these elements in cancer is very much an active area of research. The extent to which retrotransposable elements contribute to cancer development may vary depending on the specific element, its location within the genome, the individual’s genetic background, and the type and stage of cancer.
For example, recent studies from my lab highlight both pro-tumour and anti-tumour effects of a single human endogenous retrovirus copy in squamous lung cancer, as well as antibody reactivity against viral envelope glycoproteins in lung adenocarcinoma, particularly in the context of immunotherapy.
Retrotransposable elements have long been implicated in the development of cancer, although the relationship is complex and not fully understood. The accumulating evidence suggests that with evolutionary time, the ability of retrotransposable elements to directly cause cancer is diminishing, whereas their co-option in pathways and mechanism that protect against cancer may be increasing.
Understanding this complex interaction may be the key to turning these old foes into friends.