What if you could get researchers, government policy-makers, pharmaceutical companies and the public together in one room to sit down and create a single shared vision for the world in 20+ years’ time where nobody gets a late stage diagnosis?
Then once we’ve all agreed on what it looks like – how do we get from here to there?

Imagine a world where our health services were more focused on maintaining our health and wellbeing than on treating disease. To some, this may sound like science fiction, but with a push towards detecting disease early – in some cases before symptoms have even appeared – it’s not as far-fetched as it may sound. And when it comes to cancer, it could make a huge difference.
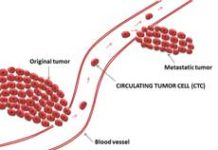
Patients who are diagnosed with cancer at an early stage –when the tumour is small and/or contained – have the best chance of long-term survival, but if cancer is detected at a late stage when the cancer has spread, its all too clear that patients’ chances are much worse. 9 in 10 patients will survive bowel cancer if it is detected at the earliest stage, but only 1 in 10 will survive when diagnosed once it has spread across the body.
The system is broken
Currently, just over half of all cancer cases are detected at the early stages, so clearly, we need to go further, faster.
One part of early diagnosis is screening, where apparently healthy members of the public are tested to see if they have cancer with no symptoms, or even signs of disease so early that cancer could be prevented from developing in the first place.
In the UK, we have phenomenal NHS screening programmes which have saved many lives. However, these only cover 3 cancer types – bowel, cervical and breast cancer – and only 6% of cancer diagnoses come through this route.
The vast majority of cancer patients are diagnosed through other routes, once they have already developed symptoms, such as though primary care. GPs work tirelessly to diagnose from early symptoms, but the tools they’ve been given aren’t perfect and the capacity of the health system in terms of diagnostic staff and technology (like MRI scanners and endoscopy) is limited.
But why is it like this?
It’s because to change the system, a variety of sectors have to come together– scientists, companies, doctors, the authorities who decide which technologies the NHS can use, government and, crucially, the public. However, all of these groups have different perspectives and do not traditionally come together. And importantly, most companies are focused on treating patients with symptoms, not on early detection.
Of course, it is crucial that we continue to work on creating and using the best treatments that we can. But ultimately, we need a new way of thinking which is built on finding disease before people even know they are sick. We desperately need innovative new technologies to detect early cancer which the health system can implement.
A roadmap for the future
Cancer Research UK set out to address these problems, with an ambition to create a huge leap forward in the way we detect cancers early. We reached out to interested parties – from pharma companies and scientists to policy makers – and for the first time, brought them all together to create a plan. That plan is our Roadmap for the Early Detection and Diagnosis of Cancer.
We consulted over 100 experts and many patients and members of the public to define a shared vision of a future where early detection of all cancers is a routine reality. The Roadmap defines the challenges that we currently face and makes a series of clear recommendations for how we can progress from here to that future. And it’s all based on united action with everyone working together to achieve progress.
A new generation of cancer detection technologies
Our Roadmap sets out a plan to develop and test a new wave of advanced technologies for detecting cancers early. We’re already seeing the development of exciting new technologies like advanced scanners, blood tests and breath tests for cancer and artificial intelligence (AI) to spot the earliest tumours in scans, for example. The Roadmap shows how these advanced technologies can best be tested and then taken up and used in the health system.
To make progress, we need to make the most of NHS and public data, which can be used to look at people’s health and spot who is at risk of cancer and should be tested. Such approaches would lead us towards a system which isn’t based solely on treating established disease, but on proactive health maintenance. All of this can only work in close partnership with the public – scientists, test companies and the government need to engage in a conversation with the public to understand what they want and will accept from data use, health monitoring and early detection.
A unified vision for a world where no-one gets diagnosed too late
We need to look further, to dream bigger, of a future world where no-one is subjected to the pain of a late-stage cancer diagnosis and the terrible outcomes that can follow. We can get there, but only by breaking down the barriers between the many groups and by uniting behind a shared vision.
At Cancer Research UK we’ve used our unique position as a trusted partner across scientists, companies, the NHS, government and the public to bring these groups together and define this future.
But we can’t do this alone, we need Government to act. The upcoming spending review is a significant opportunity for the Government to invest in more diagnostic staff and equipment, which is absolutely crucial if we are to diagnose more patients early. We also need to invest in developing the new technologies that we need and to overhaul the system to get these new approaches implemented quicker.
We have the plan, now we need action. We will continue to work to keep these groups together and work through the recommendations made in the Roadmap, to create that world where no more lives are lost due to a diagnosis that came too late.
None of this will be possible without government investment, or without you – our supporters and the public. You must be at the heart of everything we plan, and through your continued support, we’ll get there.
David Crosby is the Head of Prevention and Early Detection Research at Cancer Research UK. To hear more about this, listen to his interview on “You, Me and the Big C” from BBC Radio 5 Live.