In a phase II DESTINY-Lung01 study, treatment with trastuzumab deruxtecan showed durable anticancer activity in 91 patients with HER2-mutated non-small cell lung cancer (NSCLC). A confirmed objective response occurred in 55% of the patients, the median duration of response (DoR) was 9.3 months, median progression-free survival (PFS) was 8.2 months, and median overall survival (OS) was 17.8 months. The safety profile included interstitial lung disease that was fatal in 2 cases. The study results support the clinical benefit of trastuzumab deruxtecan in this setting, a clinical context in which no targeted agents are currently approved. The findings are published by Dr. Bob T. Li of the Thoracic Oncology and Early Drug Development Service, Memorial Sloan Kettering Cancer Center in New York, NY, US and colleagues in the 20th January 2022 of The New England Journal of Medicine.
The authors wrote in the study background that HER2 mutations drive approximately 3% of non-squamous NSCLC and are associated with female sex, never-smoking history, and a poor prognosis, as well as with a slightly younger age and higher incidence of brain metastases. Patients with HER2-mutated NSCLC are currently treated with standard chemotherapy or immunotherapy, which have limited activity in second- or later-line treatment.
Clinical studies of trastuzumab in combination with chemotherapy showed disappointing outcomes in patients with NSCLC because of difficulties in defining precise molecular criteria for the selection of patients who would benefit from HER2-targeted agents and the rarity of high-level HER2 protein expression or HER2 amplification in NSCLC in comparison to breast and gastric cancers. Efforts in targeting HER2-mutated NSCLC with HER2 tyrosine kinase inhibitors (TKIs) and HER2 antibody–drug conjugate trastuzumab emtansine have produced encouraging, but inconsistent results.
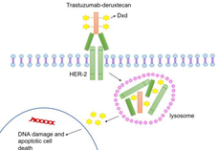
Trastuzumab deruxtecan is an antibody–drug conjugate consisting of a humanised anti-HER2 monoclonal antibody linked to a topoisomerase I inhibitor payload through a tetrapeptide-based cleavable linker. Results from first-in-human study with trastuzumab deruxtecan in patients with HER2-mutated NSCLC showed preliminary antitumour activity in 11 patients with a confirmed objective response in 72.7% of the patients.
Furthermore, translational studies showed that HER2-mutated NSCLC may preferentially internalise the HER2 receptor antibody–drug conjugate complex regardless of HER2 protein expression and overcome resistance to other HER2-targeted agents, which supports the further development of trastuzumab deruxtecan in this population.
DESTINY-Lung01 investigators conducted a multicentre, international, phase II study with trastuzumab deruxtecan administered to patients with metastatic HER2-mutated NSCLC refractory to standard treatment. The primary outcome was objective response assessed by independent central review. Secondary outcomes included the DoR, PFS, OS, and safety. Biomarkers of HER2 alterations were assessed.
In total, 91 patients were enrolled. The article published in The New England Journal of Medicine elaborates the results from the primary analysis involving the fully enrolled cohort of patients with HER2 mutations. The median duration of follow-up was 13.1 months (range, 0.7 to 29.1).
Centrally confirmed objective response occurred in 55% of the patients (95% confidence interval [CI] 44 to 65). Responses were observed across different HER2 mutations, as well as in patients with no detectable HER2 expression or HER2 amplification.
The median DoR was 9.3 months (95% CI 5.7 to 14.7). Median PFS was 8.2 months (95% CI 6.0 to 11.9). Median OS was 17.8 months (95% CI 13.8 to 22.1).
The safety profile was generally consistent with those from previous studies. Grade 3 or higher drug-related adverse events occurred in 46% of patients with the most common adverse event being neutropenia in 19%. Drug-related interstitial lung disease occurred in 26% of patients and resulted in death in 2 patients.
The authors commented that the lack of a comparator group in this study necessitates further clinical research. In addition, patients who had previously been treated with HER2-directed antibodies or antibody–drug conjugates were excluded from the study. Whether clinical activity of trastuzumab deruxtecan is similar in such patients, as has been previously observed in breast cancer, remains to be determined.
The authors concluded that their results provide clinical evidence of antitumour activity of trastuzumab deruxtecan in patients with previously treated HER2-mutated metastatic NSCLC. However, interstitial lung disease is an important risk that requires careful safety monitoring and management.
A randomised phase II DESTINY-Lung02 study is ongoing in patients with HER2-mutated NSCLC to further evaluate the efficacy and safety of trastuzumab deruxtecan, including a lower dose of 5.4 mg per kilogram which is the recommended and approved dosage in HER2-positive breast cancer. DESTINY-PanTumor01 study is also under way to evaluate trastuzumab deruxtecan in targeting HER2 mutations across other cancer types.
Drs Antonio Passaro and Solange Peters wrote in an accompanied editorial that trastuzumab deruxtecan is changing the treatment for all HER2-mutated NSCLC, for which the antibody–drug conjugates appear clinically more effective than TKIs. A well-structured plan is already shaping the future of personalised antibody–drug conjugate medicine, including further improvements in drug design, the search for biomarkers predictive of efficacy or responsible for acquired resistance, and the development of combination strategies.
The DESTINY-Lung01 study was supported by Daiichi Sankyo and AstraZeneca. Dr. Li was also supported by grants from the US National Institutes of Health to the Memorial Sloan Kettering Cancer Center.