In a phase II part of a phase I/II study that enrolled patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) who had received at least two lines of treatment previously, a fixed course of glofitamab therapy led to a complete response (CR) in 39% of the patients. Responses were observed early, usually at the first scheduled response assessment at approximately 1.4 months. Responses were durable, with 78% of the patients with a CR continuing to have remission at 12 months. The estimated 12-month overall survival of 50% was meaningful given the poor prognosis with conventional chemotherapy in this disease. Long-term follow-up data from the supporting cohort of patients who had a CR at lower doses of glofitamab confirm that durable complete remissions lasting several years can be observed with this fixed-duration treatment. The findings are published by Dr. Michael J. Dickinson of the Peter MacCallum Cancer Centre in Melbourne, Australia and colleagues on 11 December 2022 in The New England Journal of Medicine. The results from a phase Ib were presented at the 64th ASH Annual Meeting (10-13 December 2022, New Orleans, US).
The authors wrote in the background that R-CHOP is the standard first-line treatment for DLBCL. However, 35 to 40% of patients have a relapse or have disease that is refractory to R-CHOP. Among patients with DLBCL who are considered to be eligible candidates for autologous stem-cell transplantation, 60% do not proceed to transplantation after the receipt of salvage chemotherapy or have a relapse shortly after the procedure. The prognosis is poor for patients who are unsuitable candidates for second-line treatment with aggressive salvage chemotherapy and for those who have received at least two lines of treatment previously.
Although CAR T-cell therapies appear to be the most effective for relapsed or refractory DLBCL after the receipt of at least two lines of treatment, they are not consistently available owing to logistic, geographic, or resourcing constraints. Not all patients who are selected to receive CAR T-cell infusion actually do so because of disease progression or death while awaiting treatment, and only approximately 40% of patients have durable remission with third-line CAR T-cell therapy.
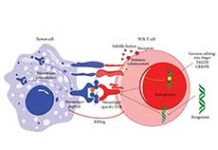
Glofitamab is distinct in the emerging class of CD20×CD3 bispecific monoclonal antibodies because it has a novel 2:1 tumour–T-cell binding configuration that confers bivalency for CD20 (B cells) and monovalency for CD3 (T cells), leading to the engagement and redirection of patients’ existing T cells to eliminate malignant B cells.
Having established a recommended phase 2 dose of glofitamab, with obinutuzumab pretreatment to mitigate cytokine release syndrome (CRS), the study team enroled expansion cohorts of patients with DLBCL who had received at least two lines of treatment previously. Pretreatment with obinutuzumab was followed by fixed-duration glofitamab monotherapy, 12 cycles in total. The primary endpoint was CR according to assessment by an independent review committee. Key secondary endpoints included duration of response, survival, and safety.
Of the 155 patients who were enroled, 154 received at least one dose of any study treatment (obinutuzumab or glofitamab). At a median follow-up of 12.6 months, 39% of the patients (95% confidence interval [CI] 32 to 48) had a CR according to independent review. Results were consistent among the 52 patients who had previously received CAR T-cell therapy, of whom 35% had a CR. The median time to a CR was 42 days (95% CI 42 to 44). The majority (78%) of CRs were ongoing at 12 months. The 12-month progression-free survival was 37% (95% CI 28 to 46).
Discontinuation of glofitamab due to adverse events occurred in 9% of the patients. The most common adverse event was CRS that occurred in 63% of the patients. Adverse events of grade 3 or higher occurred in 62% of the patients, with grade 3 or higher CRS in 4% and grade 3 or higher neurologic events in 3%.
The authors commented that in contrast to CAR T-cell therapies, the neurologic adverse events observed with glofitamab were uncommon and mostly mild. The incidence of treatment discontinuation due to adverse events was low. Most fatal adverse events (5 of 8 events) were related to COVID-19. They also wrote that the efficacy of glofitamab, its novel mechanism of action, and unique 2:1 structure provide a strong rationale for combinations with other treatments.
In an accompanied editorial, Nancy L. Bartlett of the Siteman Cancer Center, Washington University School of Medicine in St. Louis, US wrote that the sequencing of treatment with CD20×CD3 bispecific agents and CAR T-cells in patients with large-cell lymphoma will need to be defined. Under the assumption that approvals will be for third-line treatment or later lines, bispecific agents will be an excellent option for the 60% of patients in whom second-line CAR T-cell therapy fails. If longer follow-up confirms that the majority of CRs with bispecific agents are durable, on the basis of the advantages of availability, including in the community setting, and more favourable immediate and late adverse events profile, bispecific agents could be considered as the initial choice for second-line or third-line treatment. CAR T-cell therapy could be held in reserve for patients who do not have a CR or who have a relapse after a CR.
New directions include the combining of bispecific agents with standard chemotherapy in earlier lines of treatment and the evaluation of new tumour targets, such as CD19 and CD22, and new immune effector targets such as natural killer cells. Treatment with T-cell–engaging bispecific agents will improve outcomes for patients with lymphoma and, once again, will show the extraordinary potential of the immune system in the treatment of cancer according to the editorialist.
In another editorial that featured the expanding role of bifunctional antibodies, Dr. Dan L. Longo wrote that glofitamab differs from other anti-CD20×anti-CD3 antibodies in that it has two anti-CD20 binding domains; it is therefore bivalent for the tumour antigen and monovalent for the T-cell CD3 protein. Whether the bivalency is a strength, or a weakness cannot be ascertained without a head-to-head comparison with the monovalent antibody mosunetuzumab. Talquetamab targets CD3 on T cells and a protein that is highly expressed on malignant plasma cells.
The study was supported by F. Hoffmann–La Roche.
References
Dickinson MJ, Carlo-Stella C, Morschhauser F, et al. Glofitamab for Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med 2022;387:2220-2231.
Bartlett NL. Bispecific Antibodies in Lymphoma — Another Win for T Cells. N Engl J Med 2022; 387:2285-2286.
Longo DL. The Expanding Clinical Role of Bifunctional Antibodies. N Engl J Med 2022; 387:2287-2290.