, by NCI Staff
To become cancerous, a cell must accumulate genetic changes that allow it to grow uncontrollably. But not all cells with these changes will actually give rise to tumors. Many things, including the immune system, can stop a single cancer cell from growing into a full-fledged tumor.
New research suggests that whether a cell with cancer-related genetic changes will go on to develop into a tumor also depends on that cell’s location in the body.
Virtually all cells in the body have the same genes. But how those genes behave can vary, depending on several factors, including where that cell lives in the body. Influenced by what are sometimes called positional gene expression programs, the varied activity of genes ensure that your nose grows on your face and your toes on your feet.
Each of these programs requires a slew of proteins and other molecules that are unique to their location. In studies in zebrafish and human cells, researchers have now found that certain oncogenes—genes that, when altered in certain ways, can cause cancer—caused a rare type of melanoma to develop in certain parts of the body due to the positional programs they could harness at those locations. The results were published March 30 in Nature.
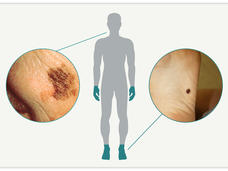
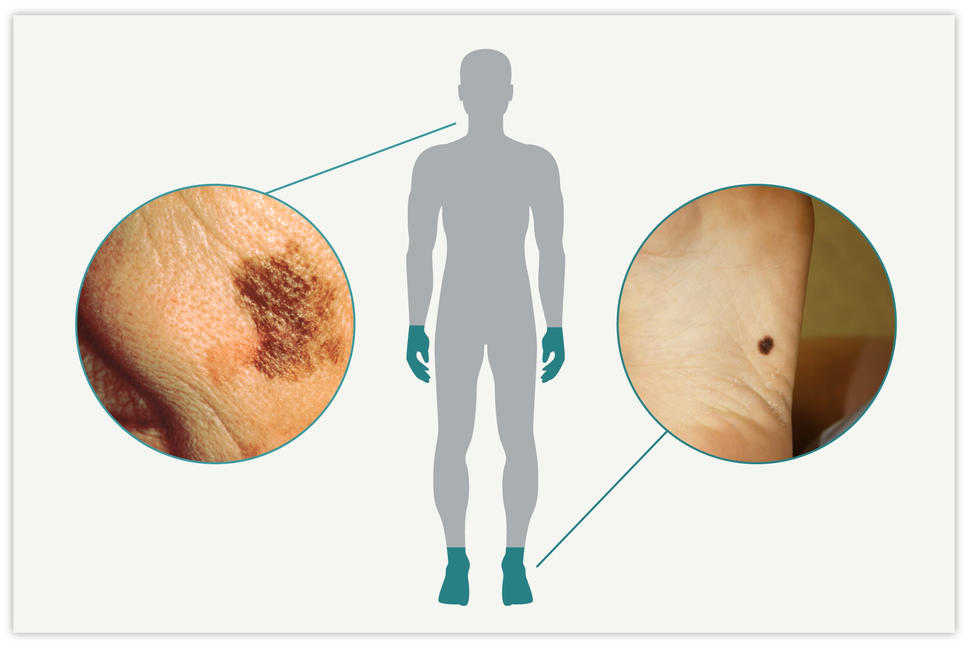
The new studies looked at acral melanoma, which grows on the palms of the hands, the soles of the feet, and under the nails. According to Richard White, M.D., Ph.D., of Memorial Sloan Kettering Cancer Center, who led the study, the findings have implications for the treatment of this rare cancer. But they may also hold clues to the way other cancer types develop and can be treated, Dr. White said.
“The idea that there may be specific [treatments] that work in one part of the body but not others is very compelling,” he said.
Same cells, different cancers
Melanoma starts in melanocytes, cells that produce and contain a pigment called melanin. This pigment helps protect the skin from damage from the sun’s ultraviolet rays. The more melanin a person has, the darker their skin is.
In addition to the skin, melanocytes are found in the eyes and even the gut. As a result, melanoma can arise in many different parts of the body. The most common type of melanoma, called cutaneous melanoma, starts in melanocytes in the skin covering central regions of the body, such as the face, chest, or back.
Over the last decade, the treatment of advanced cutaneous melanoma has been revolutionized by the discovery that many such cancers are driven by a set of genes in a cellular communication network called the MAPK signaling pathway. Drugs that target this pathway are now part of standard treatment for people with advanced cutaneous melanoma.
But noncutaneous melanomas, such as those that form in the body’s extremities (the acral regions), usually aren’t driven by alterations in the MAPK pathway.
“The [best] current melanoma therapies, which were designed to treat cutaneous melanoma, don’t work as well in acral melanoma,” explained Christine Nadeau, Ph.D., of NCI’s Division of Cancer Biology, who was not involved with the new study.
Although acral melanoma is rare overall, it’s more common in Black, Hispanic, and Asian people than in White people. It’s perhaps best known for causing the untimely death of the singer Bob Marley, at the age of 36.
This higher incidence in minority populations and fewer treatment options likely contribute to poorer survival after a melanoma diagnosis in these groups, said Dr. Nadeau. “And acral melanoma has been understudied,” added Dr. White.
In their study, his team used different techniques to look for genetic differences between acral and cutaneous tumor samples from people with one of these types of melanoma. The analysis allowed them to identify the presence of specific genetic changes, such as mutations in specific cancer-related genes, and analyze the behavior of specific genes—that is, which genes were active and influencing cellular behavior.
As expected, mutations in BRAF, a gene in the MAPK pathway, were the most common mutations in the cutaneous melanoma samples. In contrast, changes in several other genes, including a known cancer-related gene called CRKL, were more common genetic changes in the acral melanoma samples.
Human limbs to fish fins
To test the cancer-causing potential of these gene changes, Dr. White and his team turned to a zebrafish model they developed. From a biological standpoint, fish may not seem to have many similarities to humans, but they actually have many. For example, similar genetic positional programs result in fish fins and human limbs, despite the evolutionary distance between them, Dr. White explained.
Plus, there are many advantages to studying cancer genetics in fish, he added.
“One is how fast and easy it is to test which combinations of gene changes might be driving cancer,” he explained. A single fish can lay several hundred eggs per week. In contrast, a mouse usually has less than 10 pups per litter, and gestation takes up to 3 weeks from conception to birth.
“The sheer number of things we can test [in fish] is just mind blowing. In this study, we tested almost 3,000 fish with different genetic combinations” that might drive acral melanoma, he said.
When the researchers engineered melanocytes in the fish to have certain BRAF mutations, tumors formed by these cells were much more likely to be found in cutaneous sites on the body’s core. In contrast, cells engineered to have several gene changes common in human acral melanomas, including in CRKL, were much more likely to form tumors in fish fins—the analog to acral regions in people.
Additional work found that only CRKL changes appeared to be necessary for acral tumors to grow. Other oncogenes made those tumors grow faster, but they weren’t needed for a cell in the fin to turn cancerous.
Further experiments in the fish showed that the activity of other, non-cancer-causing genes in melanocytes also differed substantially depending on where in the body the cells resided, such as the core versus the fins. Many of the most active molecular pathways in fin melanocytes were involved in limb development.
Similar results were seen in acral melanoma samples taken from people, with many of the most active gene-expression pathways related to limb development.
Hijacking a positional pathway
For the team, the next obvious question was: Are the cancer-causing gene changes seen in acral melanoma, like CRKL changes, somehow exploiting positional programs found only in the fins and limbs? And how?
One such program, controlled by a group of genes known as HOX13, was highly active in almost all the samples taken from patients’ acral melanomas but less active in samples taken from cutaneous melanomas. In the fish, an excess of the protein produced by CRKL ramped up this pathway and caused runaway cell growth. But blocking the HOX13 pathway slowed tumor growth in the fins, regardless of CRKL activity.
Since CRKL appears to need this positional pathway to drive tumor growth, a kind of natural selection is likely at play in melanoma, Dr. White explained. CRKL alterations aren’t any more likely to occur in the skin on the hands and feet than in skin on other parts of the body. But only in the hands and feet can they express their oncogenic potential, so to speak, and cause runaway cell growth.
These results go beyond what was already known about how cancer cells are influenced by the normal cells around them, also called the tumor microenvironment, said Dr. Nadeau. “It’s about more than just where the cells are—it’s about them participating in these developmental programs that are necessary for things like limb development,” she explained.
The findings may not only help researchers better understand the underlying biology of these cancers, Dr. White explained, but may also be relevant to developing treatments.
“We think these [positional pathways] are actually therapeutic vulnerabilities,” he said. That is, they raise the possibility that a tumor can be treated by targeting the cross talk between an oncogene and the positional programs in a cell that tell it where it is in the body. “If you can find a drug that cuts off that communication, is that enough to prevent the cell from forming a tumor?”
The concept of cancer cells’ dependence on a positional program leads to many other interesting questions, said Dr. White, such as whether this phenomenon is found in other types of cancer besides melanoma. He thinks it may be, noting that researchers have already found other examples of tumors that behave differently based on their location, such as tumors on the left or right side of the colon, or the head or the tail of the pancreas.
This concept also raises the intriguing idea of treating metastatic cancer differently based on which organ cancer cells have spread to, he added. “Maybe we should treat liver metastases different from brain metastases or lung metastases or kidney metastases,” he said.
Another interesting question raised by the current study is whether the locations in the body to which different tumor types preferentially spread, or metastasize, are related to the positional programs expressed at those locations, added Dr. Nadeau.
Such questions will need to be answered with future research. For now, said Dr. Nadeau, a key result from the current study is the identification of a group of genes—led by CRKL—that help drive acral melanoma and are distinct from those involved in cutaneous melanoma.
“This is important, because identifying early genetic features of cells that go on to become acral melanomas can help provide biomarkers for earlier detection and help with the design of new treatments,” she said. “Right now, patients with [advanced] acral melanoma have limited treatment options. It’s quite likely that something in the CRKL pathway might be a target for treatments to stop tumor growth.”