When cancer is diagnosed, doctors will assign the cancer a stage.
Typically, this ranges from stage 1, meaning the cancer is small and hasn’t spread beyond where it first started, to stage 4, meaning the cancer has spread from where it started to another body organ.
But there’s also a stage before these that refers to the presence of abnormal cells that are what we call pre-cancerous. This is called stage 0.
At this stage, the number of abnormal cells is too small to form a tumour, but it may develop into cancer at some point.
Ductal carcinoma in situ (DCIS) is an example of stage 0 cell changes. In DCIS, some cells in the lining of the ducts of breast tissue have started to become abnormal but have not started to spread to surrounding breast tissue.
Whilst DCIS itself isn’t invasive, it is often treated with surgery to remove all or part of the breast as it has the potential to develop into invasive breast cancer. This surgery may be followed by radiotherapy to remove any remaining abnormal cells.
However, not all cases of DCIS will develop into cancer, meaning that some people receive unnecessary treatment, known as overtreatment.
Currently, there is no way of telling whether DCIS will develop into invasive breast cancer, which is why most cases are treated in the same way.
New research from a Cranfield-Exeter University collaboration, working as part of the Cancer Grand Challenges PRECISION team, may have found a way we can predict which cases could develop into invasive breast cancer and thus require treatment, reducing the burden of overtreatment in DCIS in future.
‘Immortalising’ the environment
To do this, the team analysed microcalcifications.
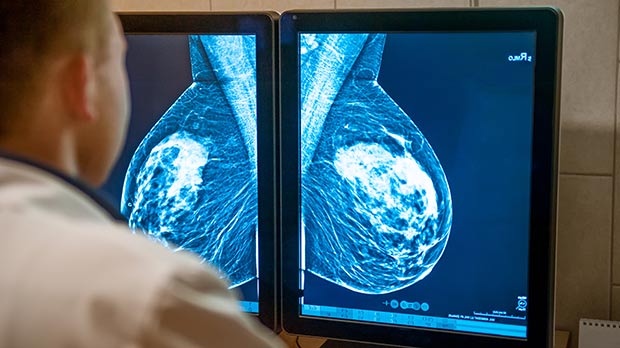
Microcalcifications are deposits of calcium salts in breast tissue that form as tiny crystals. They don’t cause any symptoms and are too small to be felt, but they can be seen as small, white specks on a mammogram.
The presence of microcalcifications on a mammogram isn’t necessarily an indication of cancer, but clusters of them could indicate cancerous or pre-cancerous changes to cells.
This is because microcalcifications don’t form without a stimulus, i.e., something in the environment of the surrounding breast tissue must change to trigger their formation, though exactly how they form remains a mystery.
But depending on what the environmental changes are, these microcalcifications can form differently.
For example, they might be made up of slightly different things alongside the calcium, which will make their structure, and therefore appearance under the microscope, slightly different.
“The primary material that these calcifications are made of is quite a fascinating material. In a way it acts like a sponge,” explains Professor Keith Rogers, Professor of Material and Medical Science at the Cranfield Forensic Institute.
“So, anything in the environment has an opportunity to be absorbed into the structure, which we can subsequently measure.”
Microcalcifications can therefore act as a snapshot of what the surrounding environment is like when they formed. Or, as Rogers puts it, the tissue environment is ‘immortalised’ in the calcifications.
When breast cancer develops, we know that the microenvironment around the cells changes. For example, the environment becomes hypoxic, meaning it lacks oxygen, and that causes it to become more acidic.
And these changes will impact on the composition of any microcalcifications that form.
That means we can compare the structure and composition of microcalcifications in people with and without breast cancer and determine how the tissue environment when they formed differed.
That can, in theory, give us clues as to whether future cases of DCIS are going to become invasive. If the calcifications look similar to those found in cases where breast cancer developed, it might be more likely to occur.
Structure and composition
The research team studied these microcalcifications in two groups of people: people with DCIS that hadn’t developed into invasive breast cancer within 5 years, and people with DCIS that had developed into cancer within 5 years.
The team mainly used a technique called X-ray diffraction, a method of determining the structure and composition of a crystal by firing a beam of X-rays at it and measuring the directions the beam is diffracted to.
They also took images of the crystals with an electron microscope.
“Our primary aim was to be able to differentiate between patients who had had DCIS, without having any kind of recurrence, and those that had recurrence after the initial DCIS diagnosis,” says Dr Sarah Gosling, a postdoctoral research associate at Keele University who led the study during her PhD at Cranfield University.
“So, we were looking at the calcifications, what they’re made of, the structure of the things that they’re made of, and whether there are differences that we can identify between these two.
“The exciting bit of the research is that we did find differences.”
The first difference they found was the size of the microcalcifications, with crystals in the invasive breast cancer group being on average 33% larger compared to the non-invasive group.
And what’s more, the composition of the crystals differed too.
An unusual addition
The main compound in the crystals of both groups was something called hydroxyapatite (HAP), a mineral similar in composition to our bones. However, in cases where the DCIS became invasive, the microcalcifications were what we call more ‘mature’ crystals with a higher crystallinity.
“Crystallinity is basically the level of order within the crystal. A crystal has high crystallinity if it’s really regular,” Gosling explains.
“So, if you have a unit in the crystal and the units next to it are all identical, that’s high crystallinity.”
“Crystallinity is then a measure of crystal maturity as well. So, the more mature the crystal, the more crystalline it tends to be.”
Crystals with high crystallinity are more stable whereas lower crystallinity crystals are easier to dissolve, and they are reabsorbed by the surrounding tissue.
But the crystals weren’t entirely HAP.
The researchers definitively found, for the first time, that the microcalcifications also contained an unusual compound called whitlockite, and that in cases where DCIS became invasive, there was a much less of it in the crystals.
Whitlockite is similar in structure to HAP but is made up of slightly different things. It is a rare mineral, and curiously, it has been central to the argument that water once existed on Mars.
The whitlockite of breast tissue contains the element magnesium and requires a relatively high concentration of it to form.
We know that magnesium levels are depleted in the early stages of breast cancer development, which causes an inflammatory response that tumours use to form new blood vessels.
When magnesium is depleted, less whitlockite can form, which may explain why there’s less of it present when DCIS becomes invasive.
However, it remains unclear whether the crystals are indicative of environmental changes that suggest DCIS will become invasive, or whether the formation of the crystals themselves are the trigger.
It could be that this process is cyclical. Changes to the microenvironment cause microcalcifications to form, and the formation of the microcalcifications causes further changes to the microenvironment, which may stimulate DCIS to become invasive.
Whether this is the case or not is something we need more research to answer.
What’s next?
While the findings of this research are exciting, it may take a while for them to translate to uses in the clinic, as most of these features of the crystals can’t be measured by standard hospital laboratory equipment.
But by knowing what features of a microcalcification might indicate that a person’s DCIS is more likely to progress into invasive breast cancer, or perhaps more importantly, that it isn’t likely, we can make better, more accurate treatment decisions for them.
People at a higher risk that their DCIS will progress can be treated appropriately with surgery, while those at lower risk can continue to be monitored without overtreatment.
And the implications of these findings extend beyond their potential clinical use.
“Even if you can never move this directly into the clinic, what you’ve got is system of biomarkers that you could use wherever you have calcification, and that could go beyond breast tissue,” says Rogers.
“There are lots of pathologies that are associated with calcification like prostate and ovarian cancers, for example, which [Gosling] is working on now. We would have been less compelled to start that work if we hadn’t got positive results from the breast tissue.”
And just because these features can’t be measured now, doesn’t mean they can’t be measured in future.
“One of the advantages of doing this from a crystallographic point of view is that we’re using X-rays. And when you have a mammogram, you’re also using X-rays,” Rogers adds.
“So, in principle, the signatures that we’re measuring are already there, when you have a mammogram, it’s just that the radiologists can’t currently measure them.”
“I think this has a very, very promising future. Because even if one of those things pays off, it will be a really fantastic result.”
Jacob
Find out more about PRECISION, the Cancer Grand Challenges team taking on their lethal vs non-lethal cancers challenge