, by NCI Staff
For a second time, researchers will test an antibiotic discovered in the 1950s as a potential treatment for cancer.
The first attempt to study the drug, novobiocin, in patients with cancer was a small clinical trial conducted 30 years ago. A young woman with advanced breast cancer responded to the drug and survived for 2 years, but most of the other participants in the trial did not benefit.
Three decades later, researchers at the Dana-Farber Cancer Institute have “rediscovered” novobiocin. They found it while testing thousands of compounds against cancer cells that had become resistant to drugs known as PARP inhibitors. These drugs are used to treat patients with various cancers, including ovarian, breast, and prostate cancers.
“Lo and behold, novobiocin was one of the very top hits in our drug screen,” said Alan D’Andrea, M.D., who directs the Center for DNA Damage and Repair at Dana-Farber. Although novobiocin is no longer used to treat bacterial infections in people, the drug is still produced for veterinary medicine and remains in drug libraries for research.
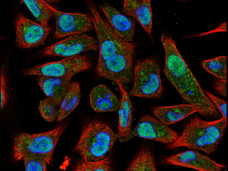
Novobiocin blocks the activity of a protein called DNA polymerase theta (Polθ or POLQ), which helps repair DNA that has become damaged as cells grow and divide. Cancer cells that cannot repair their damaged DNA die.
Polθ and the protein targeted by PARP inhibitors are both involved in a “backup” form of DNA repair. These backup pathways become particularly important when a repair process known as homologous recombination stops working, as happens in some cells with mutations in the BRCA1 or BRCA2 genes.
In a new study, Dr. D’Andrea and his colleagues found that novobiocin killed cancer cells with deficiencies in homologous recombination repair. The drug also shrank tumors in mouse models of triple-negative breast cancer that had mutations in DNA-repair genes. The researchers reported their findings in Nature Cancer on June 17.
“Most important, we found that novobiocin killed tumors that had become resistant to a PARP inhibitor,” said Dr. D’Andrea. “Novobiocin was even more effective in combination with a PARP inhibitor.”
Based on these results, his team is developing NCI-supported clinical trials to test the drug’s ability to treat cancers that have become resistant to PARP inhibitors.
The Dana-Farber team is not alone in finding that a Polθ inhibitor may have anticancer activity. Scientists from the Institute for Cancer Research in London and their colleagues have reported that a Polθ inhibitor called ART558 also can kill cancer cells and tumors that have become resistant to PARP inhibitors.
Using a Polθ inhibitor in combination with a PARP inhibitor in patients with cancers that have mutations in BRCA genes might prevent resistance from emerging in the first place, the latter group of researchers reported in Nature Communications on June 17.
The company that developed the drug, Artios, said that it plans to test the drug in clinical trials.
Trying to Solve a Pressing Clinical Problem
PARP inhibitors, including olaparib (Lynparza) and talazoparib (Talzenna), block an enzyme called poly (ADP-ribose) polymerase, or PARP, that helps repair damaged DNA. The drugs are used to treat cancers that have alterations in DNA-repair genes, namely BRCA1 or BRCA2.
Cells that have BRCA mutations are forced to rely on an alternative DNA repair process that involves PARP. As a result, these cells are particularly sensitive to PARP inhibitors and die. This is known as synthetic lethality.
BRCA gene alterations occur most often in ovarian, breast, pancreatic, and prostate cancers. PARP inhibitors have been mainstays for treating breast cancers that have BRCA mutations, and the drugs have recently been approved for treatment of prostate cancers with the mutations.
However, cancers eventually find ways to bypass the loss of PARP. “Nearly all women with ovarian cancer who are treated with PARP inhibitors will experience a relapse,” said Dr. D’Andrea. “When the tumor grows back, it is resistant to PARP inhibitors, and at this point there are no treatments—it’s a terrible clinical problem.”
In 2015, Dr. D’Andrea’s team reported that tumors that are deficient in homologous recombination repair become overly dependent for their growth and survival on POLθ.
The new study of novobiocin suggests that synthetic lethality may arise from the inhibition of Polθ in cancer cells that have defects in homologous recombination repair. The researchers cautioned that novobiocin “can overcome some but not all mechanisms of PARP resistance.”
In an editorial in Nature Cancer, Thomas Helleday, Ph.D., of the Karolinska Institute, wrote that novobiocin showed “impressive anticancer activity” in the laboratory and was “even better when used in combination with a PARP inhibitor.”
Dr. Helleday noted that although ART558 and novobiocin inhibit Polθ through different mechanisms, the results were similar. In the laboratory, ART558 killed cancer cells and shrank tumors in rats, the researchers found.
Both novobiocin and ART558 “constitute powerful tools that demonstrate the relevance of targeting Polθ in cancer,” Dr. Helleday wrote. It remains to be seen whether these drugs benefit people with cancer, he added, but the strategy of inhibiting Polθ in tumors with defects in homologous recombination appears “promising.”
A Clinical Trial through NCI’s NExT Program
In recent months, Dr. D’Andrea has worked with Geoffrey Shapiro, M.D., Ph.D., of Dana-Farber and Percy Ivy, M.D., of NCI’s Division of Cancer Treatment and Diagnosis, to develop clinical trials of novobiocin through the NCI Experimental Therapeutics (NExT) Program.
NExT provides resources to develop promising treatments that address unmet needs in cancer care and that might not be adequately supported by the private sector. As part of this general support, NCI’s Developmental Therapeutics Program will ensure that capsules containing the appropriate doses of novobiocin (the drug is given by mouth) are available for participants in the trial.
The study will test novobiocin, both alone and in combination with a PARP inhibitor, in patients whose tumors have developed resistance to PARP inhibition, according to Dr. D’Andrea. The study will primarily include patients with ovarian or breast cancers and could start enrolling patients later this year.
“We all want to better understand how to effectively treat patients whose cancers develop resistance to PARP inhibitors,” said Dr. Ivy. “I’m cautiously optimistic about novobiocin based on the outstanding preclinical data, but we have to do the study.”
The clinical trial could help researchers understand the impact of inhibiting Polθ on multiple DNA repair pathways. “We want to know whether novobiocin pushes a cancer cell beyond its ability to compensate for losing DNA repair pathways, leading to its death,” Dr. Ivy said.
How Science Happens: A Patient’s Legacy
One of the challenges in the research, Dr. D’Andrea said, has been to identify patients with cancer who are likely to respond to novobiocin. “Identifying potential responders to any cancer therapy is a challenge, and until now, we haven’t known whom to give novobiocin to,” he added.
Their efforts got a big boost from the first attempt to repurpose the drug for patients with cancer. “As we read about that research, we thought that we might be on to something and moved forward with our own study of novobiocin,” said Dr. D’Andrea.
The researchers were most intrigued by the woman with advanced breast cancer in the trial who responded to the drug after running out of other treatment options. At the time, surviving with metastatic disease for 2 years was exceptional.
“Although there was no way of knowing with any certainty, we suspected that this patient may have had a BRCA mutation,” said Joseph Paul Eder, M.D., who led the 1991 clinical trial of novobiocin. “She had the right family history and family background for a BRCA mutation.”
At the time, the BRCA genes had not yet been discovered, but the woman’s exceptional response suggested that the drug might help treat certain cancers.
Now at the Yale Cancer Center, Dr. Eder credits Dr. D’Andrea and others for identifying the target of novobiocin and demonstrating the drug’s potential as a cancer therapy. But the “chance finding” of his patient’s exceptional response 30 years ago may have played a small role in the current research, he noted.
“We always hope that no patient’s willingness to participate in a clinical trial will be in vain,” said Dr. Eder. “Here we have an example of how people who have volunteered for a clinical trial and have died from cancer can, decades later, bring us a step closer to helping others with this dreaded disease.”
Another author of the 1991 study, Lowell E. Schnipper, M.D., of Harvard Medical School agreed.
“It’s striking that when you plant a seed, it may germinate three decades later and take a form you had no idea about,” Dr. Schnipper said. “That is how science works.”